The Union Health Ministry is set to launch a nationwide Human Papillomavirus (HPV) vaccination programme targeting girls aged 14 years.
- March 4 is observed as International HPV Awareness Day.
- The WHO’s Strategic Advisory Group of Experts on Immunization (SAGE) states that the HPV vaccine is highly effective for the prevention of HPV serotypes 16 & 18, which cause 70% of cervical cancers.
- Sikkim (2018): First Indian state to provide free Gardasil HPV vaccine to girls aged 9–14 years, two doses six months apart.
About HPV Vaccination Programme
- Cost & Access: The vaccination will be voluntary and free of cost, ensuring equitable access across socio-economic groups.
- Implementation: HPV vaccination will be conducted exclusively at designated government health facilities, including the Ayushman Arogya Mandirs (Primary Health Centres), Community Health Centres, Sub-District and District Hospitals, and Government Medical Colleges.
Why Target Age 14 for HPV Vaccination?
- Optimal Timing for Protection
- Vaccines work best before exposure to HPV.
- Vaccinating at age 14 ensures protection well before potential infection.
- Supported by global and Indian studies showing strong, durable protection in this age group.
National Technical Advisory Group on Immunisation
- It is an apex advisory body to the Government of India on vaccine and immunization policy.
- It recommended introducing the HPV vaccine in the country’s Universal Immunisation Programme (UIP).
- One-time catch-up dose for girls aged 9–14 years, followed by routine vaccination starting at 9 years.
|
- Target Group: Adolescent girls aged 14 years.
- Purpose: Prevent cervical cancer, the second most common cancer among Indian women (~80,000 new cases and 42,000 deaths annually).
- Vaccine Used: Gardasil, a quadrivalent HPV vaccine, for protection from HPV types 16 and 18, which cause cervical cancer, as well as types 6 and 11.
- Dosage: Evidence supports single-dose protection in the recommended age group.
- Procurement: Secured via Gavi, the Vaccine Alliance, ensuring quality and cold-chain standards.
- Global Context: Over 90 countries now implement single-dose HPV vaccination, showing reductions in HPV infection and cervical cancer incidence.
- National Technical Advisory Group on Immunisation: India’s approach is grounded in global best practices, national disease burden evidence, and expert recommendations of the National Technical Advisory Group on Immunisation.
Types of HPV Vaccines
- Quadrivalent (Gardasil): Protects against HPV 16, 18, 6, 11 (6 & 11 cause genital warts).
- Bivalent (Cervarix): Protects against HPV 16 & 18.
- Nonavalent (Gardasil 9): Protects against 9 HPV strains.
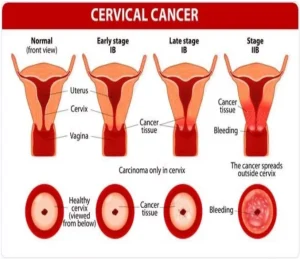
About Cervical Cancer
- About: Cervical cancer is a growth of cells that starts in the cervix.
- The cervix is the lower part of the uterus that connects to the vagina.Causes: It is caused by persistent infection with the human papillomavirus (HPV).
About Human Papillomavirus
- Human papillomavirus (HPV) is a viral infection that’s passed between people through skin-to-skin contact.
- There are over 100 varieties of HPV, more than 40 of which are passed through sexual contact and can affect genitals, mouth, or throat.
- However, some types of HPV can lead to the development of genital warts and even cancers of the cervix, anus, and throat.
|
- Risk Factors: Unprotected sex, multiple sexual partners, smoking, hormonal contraceptives, STDs.
- Screening Tests: the Papanicolaou (Pap) test and the HPV screening.
- The pap test looks for abnormal changes in cervical cells, while the HPV screening checks for the virus that can cause these changes.
- Prevention:
- Vaccination at age 9–14 years is a very effective way to prevent HPV infection, cervical cancer and other HPV-related cancers.
- Cervical Cancer in India:
- Cervical cancer is the second most common cancer in Indian women.
- India accounts for nearly a fifth of global cervical cancer cases, with approximately 1.25 lakh cases and 75,000 deaths annually.
 According to a recent study published in The Lancet, India accounted for nearly one in every four deaths globally due to cervical cancer.
According to a recent study published in The Lancet, India accounted for nearly one in every four deaths globally due to cervical cancer.
Cervavac: India’s Indigenous HPV Vaccine
- Quadrivalent Nature: Cervavac is India’s first indigenously developed quadrivalent human papillomavirus (qHPV) vaccine designed to prevent cervical cancer.
- It is based on VLP (Virus-Like Particles)
- Developed by: The Serum Institute of India in collaboration with the Government of India’s Department of Biotechnology (DBT).
- How does the Vaccine Work?
- HPV vaccines prevent the entry of four common HPV types: 16, 18, 6, and 11. Key HPV Types Targeted:
- Types 16 and 18: These are the most oncogenic HPV types, causing approximately 70% of all global cervical cancer cases.
- The vaccine focuses on preventing infections from these high-risk strains.
| A quadrivalent vaccine is a vaccine that works by stimulating an immune response against four different antigens, such as four different viruses or other microorganisms. |
Challenges in Cervical Cancer Control
- High Disease Burden: Cervical cancer remains high in Asia and Africa. Many countries have incidence and mortality rates above WHO elimination thresholds.
- Geographical and Socioeconomic Inequalities: Significant disparities exist globally; higher rates in countries with lower human development.
WHO Global Strategy to Eliminate Cervical Cancer (2022)
- Vaccination Target: 90% of girls will be fully vaccinated with HPV vaccine by age 15 by 2030.
- Screening Target: 70% of women screened with a high-performance test between ages 35–45.
- Goal: Early detection and treatment of pre-cancerous lesions.
- Treatment Target: 90% of women identified with cervical disease to receive appropriate treatment.
|
Way Forward
- Expand Screening and Early Detection: Increase access to high-performance screening (Pap test, HPV test) for women aged 35–45.
- Coordinated Campaigns: Requires coordinated campaigns by:
- Government
- Doctors and healthcare providers
- Ground-level health workers
![]() 25 Feb 2026
25 Feb 2026

 According to a recent study published in The Lancet, India accounted for nearly one in every four deaths globally due to cervical cancer.
According to a recent study published in The Lancet, India accounted for nearly one in every four deaths globally due to cervical cancer.