GLP-1 therapy, long used for diabetes management, has recently surged in popularity due to its expanded role in obesity and weight management.
- Patent expiry (March 2026): The patent of Novo Nordisk’s semaglutide widely used in GLP-1 therapy expired in India, ending its monopoly and enabling the entry of cheaper generic versions.
Best Online Coaching for UPSC
About (Glucagon-Like Peptide-1 (GLP-1) Therapy
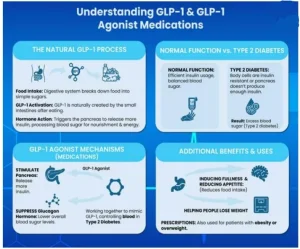
- GLP-1 (Glucagon-Like Peptide-1) therapy refers to the use of incretin-based drugs that mimic the natural GLP-1 hormone to manage type-2 diabetes and obesity.
- Incretin-based drugs are a class of medications for type 2 diabetes
- Working Mechanism: These drugs act by enhancing glucose-dependent insulin secretion, suppressing glucagon release, delaying gastric emptying, and reducing appetite, thereby improving glycaemic control and promoting weight loss.
- Initially developed for diabetes management, GLP-1 therapy has gained prominence as an effective pharmacological intervention for obesity and metabolic syndrome.
- It is particularly relevant in the Indian context due to the high prevalence of insulin resistance and the “thin-fat phenotype.”
What are GLP-1 Drugs?
- GLP-1 (Glucagon-Like Peptide-1 agonist) drugs are prescription medications used to treat type 2 diabetes and obesity.
- For Example: Semaglutide injection, Tirzepatide, Dulaglutide
What is Diabetes ?
- Diabetes is a chronic metabolic disease characterized by high levels of glucose (sugar) in the blood due to problems in insulin production, insulin action, or both.
- Insulin is a hormone produced by the pancreas that helps cells absorb glucose for energy.
Types
- Type 1 diabetes is a chronic autoimmune condition where the immune system destroys insulin-producing beta cells in the pancreas, leading to little or no insulin production.
- Type 2 diabetes stops the body from using insulin properly.
- Family history of diabetes, obesity/overweight and not getting enough exercise increases the risk of getting type 2 diabetes.
|
- The first GLP-1 medication was approved by the United States’ Food & Drug Administration in 2005.
- Prescription Guideline: In India, GLP-1 drugs can only be prescribed by endocrinologists, internal medicine specialists, and cardiologists.
- They cannot be purchased over the counter.
- They are included in the WHO Essential Medicines List for diabetes management.
- Regulatory Regime: To ensure ethical pharmaceutical practices in the supply chain of GLP-1, the Drug Controller General of India has intensified its regulatory surveillance against the drug’s unauthorised sale and promotion.
Click to Explore UPSC Offline Coaching
Implications of Semaglutide Patent Expiry
- Improved affordability: Entry of generics has significantly reduced prices, making GLP-1 therapy accessible to a larger population.
- Enhanced accessibility: Wider availability across India, especially benefiting middle- and lower-income groups.
- Increased market competition: Multiple pharmaceutical players entering the segment, leading to innovation and price efficiency.
- Boost to public health outcomes: Greater access can aid in tackling the rising burden of obesity, diabetes, and related metabolic disorders.
- Regulatory challenges: Increased risk of misuse, over-prescription, and misleading advertisements necessitating stricter oversight.
- Healthcare system impact: Potential reduction in long-term disease burden and associated healthcare costs.
- Shift in treatment landscape: Strengthens pharmacological management of obesity, bridging the gap between lifestyle changes and bariatric surgery.
What is the “Thin-Fat Phenotype”?
- The thin-fat phenotype refers to a condition where individuals appear thin by Body Mass Index (BMI) but have high body fat percentage, especially visceral (abdominal) fat.
- Such individuals often have:
- Low muscle mass
- High fat deposition around organs
- Increased insulin resistance
- Common in populations like Indians and South Asians
- Leads to higher risk of:
- Type-2 diabetes
- Cardiovascular diseases
- Metabolic syndrome
|
![]() 10 Apr 2026
10 Apr 2026